This study explores the combined effects of marine n-3 polyunsaturated fatty acids (PUFA) and ɤ-linolenic acid (GLA) as anti-inflammatory agents to help in the treatment of inflammatory disorder of rheumatoid arthritis.
Shifting the Pathological Changes of Rheumatoid Arthritis
Rheumatoid arthritis (RA) is one of the most common autoimmune disorders. The main 
Numerous randomized controlled clinical trials have reported a strong anti-inflammatory potential of marine n-3 long-chain polyunsaturated fatty acids (n-3 LC-PUFAs) and improved clinical parameters of RA. These effects are mostly attributed to eicosapentaenoic acid (EPA, C20:5 n-3) and docosahexaenoic acid (DHA, C22:6 n-3) acting in two ways.
- Firstly, they interfere with the enzymatic conversion of arachidonic acid (AA, C20:4 n-6) to pro–inflammatory prostaglandins (PGs) and leukotrienes (LTs).
- Secondly, EPA is a direct precursor in biosynthetic pathway of anti-inflammatory PGs (series-3) and LTs (series-5). Dietary n-3 LC-PUFAs replace AA in the phospholipid bilayer of the cell and then alter the membrane composition and fluidity, as well as cell signalling, gene transcription and metabolism of proresolving mediators.
- Specialized proresolving mediators (resolvins, protectins and maresins) are bioactive lipid regulators implicated in the termination of inflammation by acting through specific G protein-coupled receptors.
- The resolution of inflammation is mediated by the termination of neutrophil recruitment, stimulation of apoptotic cells phagocytosis by macrophages, and their subsequent leaving of inflammatory tissue.
- These novel lipid mediators provide rationale for further research on the beneficial effects of fish-oil enriched diets.
Likely other n-6 PUFAs, ɤ-linolenic acid (C18:3n-6, GLA), found mostly in borage, black current seed and evening primrose oil (EPO), exerts anti-inflammatory activity. Namely, it can be converted to dihomo-ɤ-linolenic acid (C20:3n-6; DGLA) and further to PGs series-1, another group of eicosanoids with potential anti-inflammatory and immunoregulatory effects.
Although these oils also contain linoleic acid (C18:2 n-6, LA), a precursor of AA, and some dietary GLA can eventually form AA, studies have shown that anti-inflammatory effects of EPO intake is prevailing. Thus, GLA may also alleviate inflammation and disease severity in patients with RA, as shown in clinical trials using high doses of 1400–2800 mg GLA per day.
Is Fish Oil Alone Enough for Rheumatoid Arthritis?
Although many studies have addressed fish oil supplementation in RA patients, the results are inconsistent. Large doses of at least 2.7 g EPA + DHA are likely required to achieve desirable outcomes in RA, but these doses can increase the risk of bleeding. In this study, RA patients were supplemented either with n-3 PUFA or n-3 PUFA and GLA to examine possible anti-inflammatory effects and clinical benefits compared to the control group of patients, who had no supplementation. Researchers also tested the possible synergistic effect of the combined n-3 PUFA + GLA supplementation.
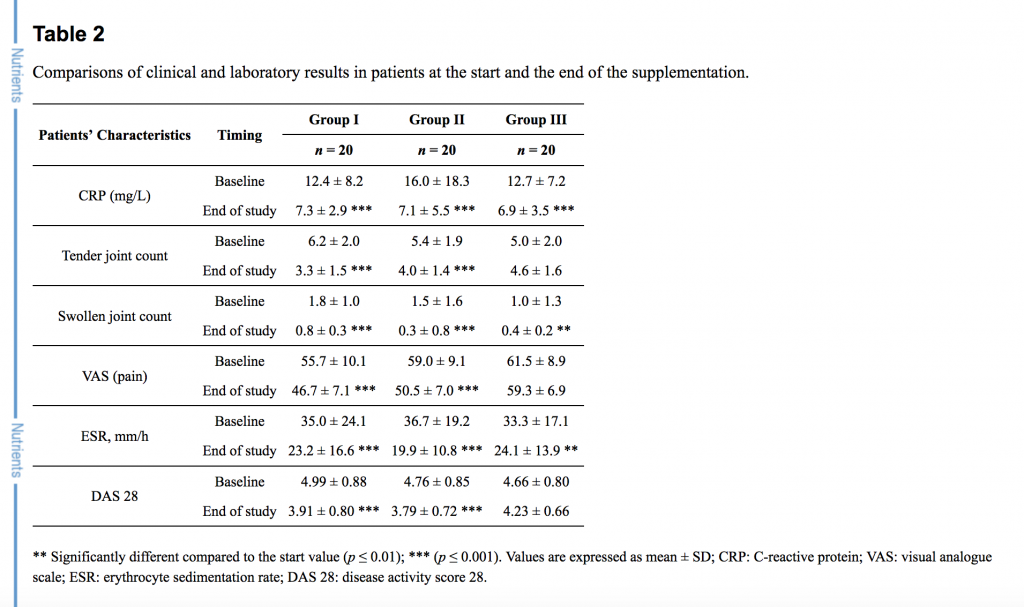
Sixty patients with active rheumatoid arthritis were involved in a prospective, randomized trial of a 12 week supplementation with fish oil (group I), fish oil with primrose evening oil (group II), or with no supplementation (group III). Clinical and laboratory evaluations were done at the beginning and at the end of the study.
The primary outcome measures were the alterations of PUFA in plasma phospholipids, with emphasis on EPA, DGLA and AA, that would indicate changes in the eicosanoids generation. The secondary outcome measure was the level of disease activity as measured with the DAS 28 disease activity score and the visual analogue scale (VAS). See Table 2 below.
Results: The Disease Activity Score 28 (DAS 28 score), number of tender joints and visual analogue scale (VAS) score decreased notably after supplementation in groups I and II (p < 0.001). In plasma phospholipids the n-6/n-3 fatty acids ratio declined from 15.47 ± 5.51 to 10.62 ± 5.07 (p = 0.005), and from 18.15 ± 5.04 to 13.50 ± 4.81 (p = 0.005) in groups I and II respectively. The combination of n-3 PUFA and GLA (group II) increased ɤ-linolenic acid (0.00 ± 0.00 to 0.13 ± 0.11, p < 0.001), which was undetectable in all groups before the treatments.
Conclusion: Daily supplementation with n-3 fatty acids alone or in combination with GLA exerted significant clinical benefits and certain changes in disease activity.