With the celebration of International Women’s Day on March 8th, it’s timely to review the differences in women’s health research and how that specifically affects investigations into treatments for overweight and obesity.
Obesity and overweight—gender similarities and differences
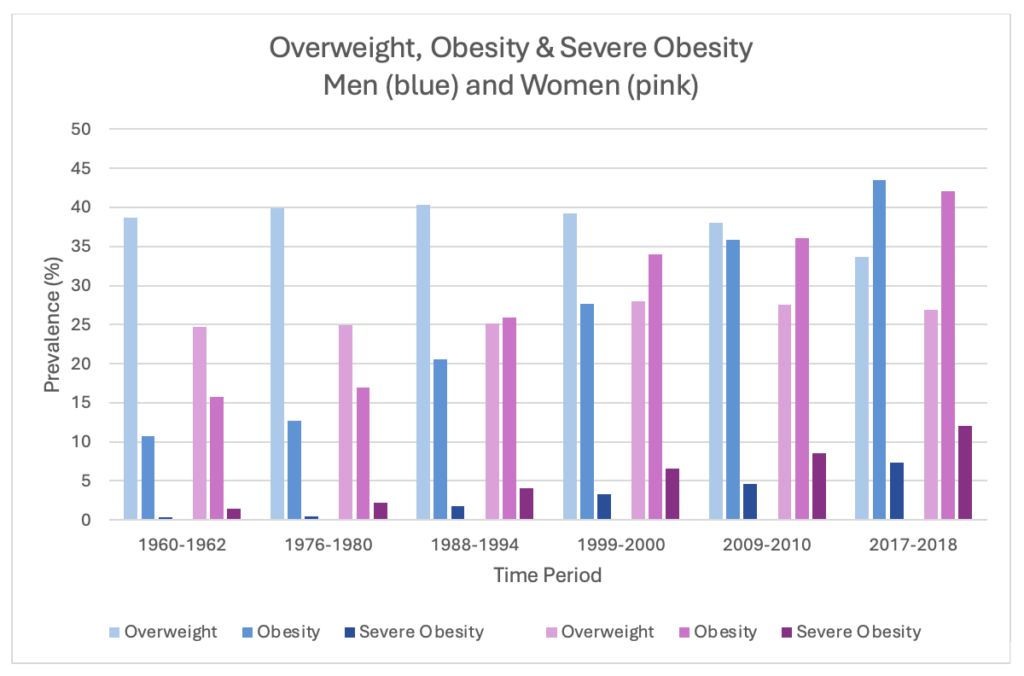
By 2022, 8 billion people over 18 (43%) were overweight globally, of whom 890 million were obese (16% of adults).1 Slightly more women* are obese than men (44% vs. 43%, respectively), and regionally, 67% of the Americas’ population is overweight.1 This reflects a trend of increasing body mass index (BMI) over the last few decades. In the USA, the Centers for Disease Control (CDC) reported that 22 states had an obesity (BMI ≥30) prevalence of 35% or more in 2022, compared to 19 states only a year before that—and no states 10 years earlier.2 While the CDC data were further broken down by race, gender differences were not analyzed.3 However, a 2020 review did break down the prevalence of overweight, obesity and severe obesity (≥40) by gender over almost 60 years, and demonstrated that women have lower rates of overweight compared to men (averaging 26.2% vs. 38.3% over the whole time period) and historically had higher levels of obesity but are now similar to men (42.1% vs. 43.5% in 2017-2018).4 In contrast, severe obesity affects more women than men (12% vs. 7.3%).4 This report also readily shows how BMI has increased over the time period studied (Figure 1).
Figure 1: Prevalence of Overweight, Obesity and Severe Obesity in the USA, 1960-20184
Recently, the likelihood of gaining weight over a 10-year period was also evaluated. After adjusting for age and race, US women had a significantly greater weight gain over 10 years compared to men, gaining about twice as much absolute weight and 2.4 times more percentage weight gain.5 The authors suggested this weight gain is likely associated with weight gains in pregnancy (said to be 1.5-2.0 kg per child) and because women are less physically active than men.5 Interestingly, the authors did not discuss weight gains associated with hormonal changes of the menstrual cycle or menopause as a cause.
We eat what we are
Could there be other reasons that women are more likely to gain significantly more weight than men in a 10-year period? Hallam et al have studied gender differences in food craving and how that affects obesity.6 Food cravings are not the same as hunger… they are a strong desire to eat, can occur with or without external stimuli and do not require “caloric need”.6 If present, external stimuli may be include visual, taste or smell cues and can elicit physiological effects such as gastric activity and salivation. Unsurprisingly, food cravings are strongly associated with increased food intake and produce long-term weight gain and lifetime high BMIs.
Hallam et al noted that there are gender differences in craving. Women experience stronger or more frequent cravings than men, whether cue-induced or not, and whether ‘in this moment’ or in general.6 Women may also be less likely to resist cravings than men and crave different types of food. Men do indeed crave more savory foods, while women typically crave sweet foods.6 In fact, 92% of those who crave chocolate are female.6 So while it’s often said “we are what we eat”, it seems it’s also true that “we eat what we are”.
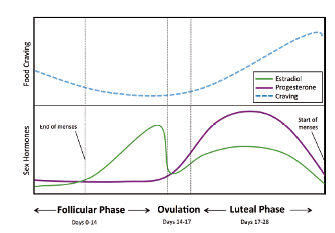
These differences in food cravings and their associated weight gain have been linked to the differences in sex hormones between men and women. Of course, sex hormones don’t just regulate menstrual cycles, fertility or body composition; they have multiple other effects including regulating energy intake and expenditure.6 In men, androgens reduce the risk of obesity and diabetes (but increase the risk in women). Estrogens reduce appetite and eating in both men and women—and reductions in estrogen increase food consumption. Menstrual cycle variations in estrogen (which decreases after ovulation) explain increased eating in the latter part of the cycle, with the strongest craving occurring just before menses (Figure 2).6 Not only do women in the later (luteal) part of the menstrual cycle eat significantly more, but they prefer sweeter foods, notably chocolate.6 A meta-analysis of 19 studies showed that during the luteal phase of the menstrual cycle women consume an average of 239 additional calories (1 MJ) a day—and up to 598 additional calories (2.5 MJ) per day.7 This amount would account for a weight gain of 10-20 pounds (4.5-9 kg) per year.6
Figure 2: Food craving during menstrual cycle changes as ovarian hormone levels fluctuate. From Hallam, et al, 2016.6 Used with permission.
Clinical research differences: “women are not small men”
While it’s apparent that women have an increased tendency gain weight over a ten-year period and are 1.6 times more likely to be severely obese than men, there have been very few studies looking at whether weight loss medications and supplements affect them differently than in men. This follows a long-standing issue in which women are underserved in health research. A McKinsey report recently identified four areas that contribute to this ‘health gap’, one of the most obvious being that the male body is the default in human biology and research.8 In fact, only 5-14% of studies in a variety of areas explicitly examine the effect of gender on outcomes.9 As Pérez and Francis succinctly note: “Women are not small men.”10 Moreover, women’s health research should not just focus on diseases that affect only women, such as those related to pregnancy, or gynecological issues, but should also address how diseases that affect both males and females may have different impacts. In addition, women’s health issues are underfunded compared to men’s.8
Gender evaluations in studies of weight loss medications
Recent studies on GLP-1 agonists for weight loss medications are no better. In the 2021 study that confirmed the success of semaglutide in weight loss in overweight and obese people, 75% of the participants were female (with an average age of 46.5) yet there were no analyses that evaluated any potential differences in its efficacy.11 Similarly, there was no such analysis in the study of tirzepatide study for weight loss, where 68% of the participants were female (aged on average 44.9).12 In 2023, Lincoff et all examined cardiovascular outcomes from semaglutide use in overweight and obese people(BMI ≥) with preexisting cardiovascular disease but without diabetes.13 They showed a 20% reduction in the risk of a composite of death from cardiovascular causes, nonfatal myocardial infarction (MI), or nonfatal stroke after an average of 34 months treatment with semaglutide compared to the placebo group—yet only 28% in the study were women.13 Given that women are twice as likely to die after an MI than men,15 it would seem sensible to have evaluated gender differences in the Lincoff trial.
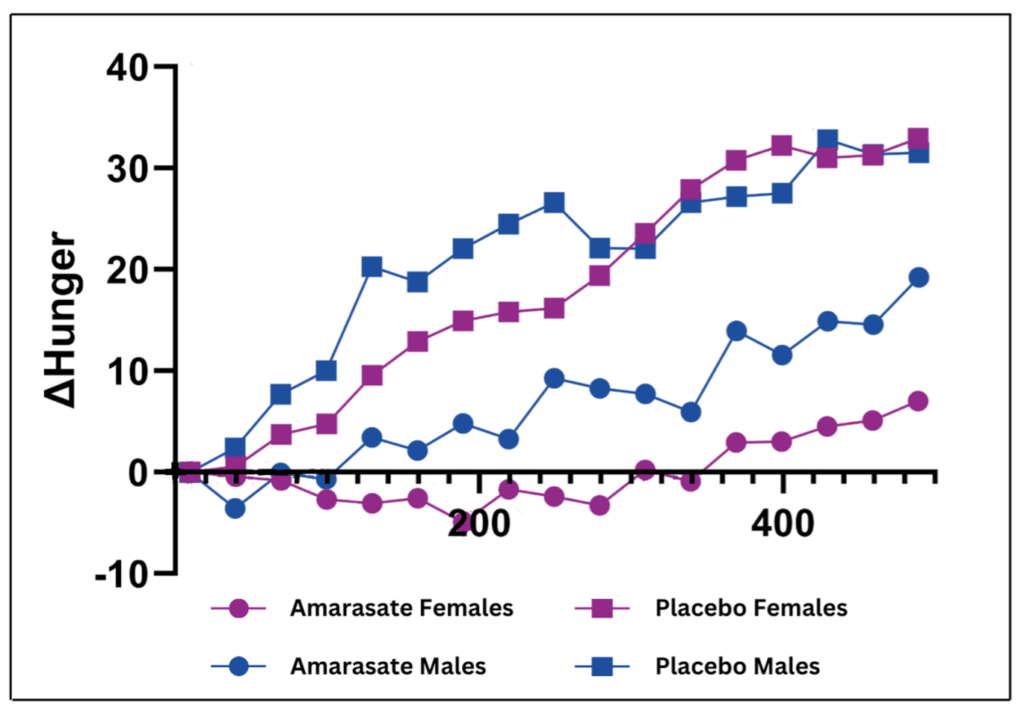
There has been one recent study that specifically evaluated the effects of a weight loss treatment on women separately from men. Walker et al examined the effect of Amarasate®, an extract of hops that has previously been shown to double the physiological GLP-1 response to eating via activation of bitter taste receptors in the gut.15 In their more recent study, Walker et al recruited 30 healthy women in a randomized, double-blind, cross-over study to evaluate how Amarasate® curbed food cravings and hunger during the last 8 hours of a 24-hour water-only fast compared to placebo.16 It took over 22 months to recruit participants, because each woman had to be fasted on the same day of her menstrual cycle for each arm of the study and on the same day of the week. As evaluated by visual analogue scales, compared to placebo, Amarasate® produced a 100% reduction in increased hunger (Figure 3, cerise)and a 120% reduction in food cravings at 24 hours—equivalent to a 30% reduction in overall hunger and a 40% reduction in overall food cravings.16 In addition, post-fast rebound eating decreased by 14.3%.16 Interestingly, a separate study that enrolled only men produced different results: men experienced an 80% reduction in increased hunger and a 25% reduction in overall hunger (Figure 3, blue).17 It seems that in these two gender-defined studies, women were more sensitive to the effects of Amarasate®.
Figure 3: Change in increased hunger determined by visual analog scale during last 8 hours of a 24-hour water-only fast in women (cerise) and men (blue).16,17
Late last month, Jill Biden announced health federal funding of $100 million for research and development into women’s health.18 Hopefully that will encourage other researchers to evaluate how gender can impact outcomes in clinical trials of interventions given to both men and women, as well as furthering the study of conditions that disproportionately affect women or have a bigger impact in women compared to men.
*For the purposes of this article, women include cis women and people assigned female at birth; men include cis men and people assigned male at birth.
References
- Obesity and Overweight. www.who.int. Published March 1, 2024. Accessed March 7, 2024. https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight#:~:text=In%202022%2C%201%20in%208
- CDC Newsroom. CDC. Published September 21, 2023. Accessed March 7, 2024. https://www.cdc.gov/media/releases/2023/p0922-adult-obesity.html
- Adult Obesity Prevalence Maps. Centers for Disease Control and Prevention. Published September 21, 2023. Accessed March 7, 2024. https://www.cdc.gov/obesity/data/prevalence-maps.html
- Fryar C, Carroll, M, Afful, J. Prevalence of Overweight, Obesity, and Severe Obesity among Adults Aged 20 and Over: United States, 1960–1962 through 2017–2018. NCHS Health E-Stats; 2020. Accessed March 7, 2024. https://www.cdc.gov/nchs/data/hestat/obesity-adult-17-18/obesity-adult.htm#1
- Tucker LA, Parker K. 10-Year Weight Gain in 13,802 US Adults: The Role of Age, Sex, and Race. Papadia FS, ed. Journal of Obesity. 2022;2022:1-10. doi: https://doi.org/10.1155/2022/7652408
- Hallam J, Boswell RG, DeVito EE, Kober H. Gender-related Differences in Food Craving and Obesity. The Yale Journal of Biology and Medicine. 2016;89(2):161-173. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4918881/
- Buffenstein R. Food Intake and the Menstrual Cycle: A Retrospective Analysis, With Implications for Appetite Research. Physiology & Behavior. 1995;58(6):1067-1077. doi: https://doi.org/10.1016/0031-9384(95)02003-9
- Ellingrud K, Pérez L, Petersen A, Sartori V. Closing the Women’s Health Gap: A $1 Trillion Opportunity To Improve Lives and Economies. McKinsey. www.mckinsey.com. Published January 2024. Accessed March 6, 2024. https://www.mckinsey.com/mhi/our-insights/closing-the-womens-health-gap-a-1-trillion-dollar-opportunity-to-improve-lives-and-economies
- Galea LA, Parekh RS. Ending the Neglect of Women’s Health in Research. BMJ. 2023;381:1303. doi: https://doi.org/10.1136/bmj.p1303
- Pérez L, Francis T. “Women Are Not Small Men”: The Global Economy Will Miss Out on $1 Trillion Annually by 2040 if We Don’t Close the Gender Health Gap. Fortune. Published January 17, 2024. Accessed March 3, 2024. https://fortune.com/2024/01/17/women-global-economy-will-miss-out-on-1-trillion-annually-by-2040-gender-health-gap-perez-francis/
- Wilding JPH, Batterham RL, Calanna S, et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. The New England Journal of Medicine. 2021;384(11):989-1002. doi: https://doi.org/10.1056/NEJMoa2032183
- Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide Once Weekly for the Treatment of Obesity. The New England Journal of Medicine. 2022;387(3). doi: https://doi.org/10.1056/nejmoa2206038
- Michael Lincoff, Kirstine Brown‐Frandsen, Colhoun HM, et al. Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes. The New England Journal of Medicine. 2023;389(24). doi: https://doi.org/10.1056/nejmoa2307563
- Women More Likely to Die After Heart Attack Than Men. www.escardio.org. Published May 22, 2023. Accessed March 9, 2024. https://www.escardio.org/The-ESC/Press-Office/Press-releases/Women-more-likely-to-die-after-heart-attack-than-men#:~:text=Prague%2C%20Czechia%20%E2%80%93%2022%20May%202023
- Walker EG, Lo KR, Pahl MC, et al. An Extract of Hops (Humulus lupulus L.) Modulates Gut Peptide Hormone Secretion and Reduces Energy Intake in Healthy-Weight Men: A Randomized, Crossover Clinical Trial. The American Journal of Clinical Nutrition. 2022;115(3):925-940. doi: https://doi.org/10.1093/ajcn/nqab418
- Walker, E.; Lo, K.; Gopal, P. Gastrointestinal Delivery of Bitter Hops Extract Reduces Appetite and Food Cravings in Healthy Adult Women Undergoing Acute Fasting. Preprints 2023, 2023090416. https://doi.org/10.20944/preprints202309.0416.v1
- Walker E, Lo K, Tham S, et al. New Zealand Bitter Hops Extract Reduces Hunger During a 24 h Water Only Fast. Nutrients. 2019;11(11):2754. doi: https://doi.org/10.3390/nu11112754
- Superville D. Jill Biden Announces $100 Million for “Life-Changing” Research and Development into Women’s Health. AP News. Published February 21, 2024. Accessed March 4, 2024. https://apnews.com/article/jill-biden-research-womens-health-arpah-shriver-149bf923d666bb980631185c0626a50e