Knee osteoarthritis (OA) is the fourth leading cause of disability. Symptoms can follow an injury, but typically they begin after age 40 of knee. Highly prevalent among obese patients, the estimated incidence is 10% to 15% in the population above 60 years of age.
The most common recommendations for managing OA are to relieve the 
Diclofenac is a well-known NSAID with anti-inflammatory, analgesic, and anti-pyretic properties, comparable or superior to other NSAIDs. The purpose of this study, published in the journal BMC Trials, was to compare the efficacy and safety of curcumin with those of diclofenac in the treatment of knee osteoarthritis (OA). Researchers also investigated the anti- flatulent and weight-lowering effects of curcumin among patients with knee OA and compared them with those of diclofenac. The researchers used a formula of curcuminoids with essential oil of turmeric to increase bioavailability.
Methods / In this randomized, open-label, parallel, active controlled clinical study, 139 patients with knee OA were randomly assigned to receive either a curcumin 500-mg (BCM-95®) capsule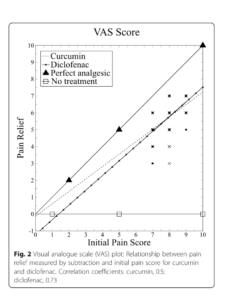
Patients underwent assessment at baseline and days 7, 14, and 28. The main outcome measure was severity of pain using visual analogue scale score at days 14 and 28. Knee Injury and Osteoarthritis Outcome Score (KOOS) (at days 14 and 28), anti-flatulent effect (at day 7), anti-ulcer effect, weight-lowering effect, and patient’s and physician’s global assessment of therapy at day 28 were included as secondary outcome measures. Safety after treatment was evaluated by recording adverse events and laboratory investigation.
Results / At days 14 and 28, patients receiving curcumin showed similar improvement in severity of pain and KOOS scale when compared with diclofenac, and the difference was not statistically significant.
- At day 7, the patients who received curcumin experienced a significantly greater reduction in the number of episodes of flatulence compared with diclofenac (P <0.01). At day 28, a weight-lowering effect (P <0.01) and anti-ulcer effect (P <0.01) of curcumin were observed.
- The dietary intake of curcumin was restricted. The patients were advised not to change their routine dietary habits and physical activity which can lead to weight gain or loss.
- None of the patients required H2 blockers in the curcumin group, and 19 patients required H2 blockers in the diclofenac group (0% versus 28%, respectively; P <0.01)
- The patients who received curcumin experienced a significantly greater reduction in body weight from baseline (P <0.01) com- pared with patients who received diclofenac (P = 0.22).
- Adverse effects were significantly less in the curcumin group (13% versus 38% in the diclofenac group; P <0.01). Patient’s and physician’s global assessment of therapy was similar in the two treatment groups.
Overall, 13% of patients receiving curcumin and 38% of patients receiving diclofenac reported at least one adverse effect (AE) and this difference was 
Conclusion / Overall, curcumin showed similar improvements in pain, stiffness, symptoms, functions of daily living, sports or recreational activities, and quality of life that have been attributed to its ability to inhibit COX-2, which results in the suppres- sion of prostaglandin synthesis. Furthermore, curcumin has been shown to suppress several pro-inflammatory cytokines and mediators of their release, such as tumor necrosis factor-alpha (TNF-α), interleukin 1 (IL-1), IL-8, and nitric oxide synthase.
Curcumin has similar efficacy to diclofenac but demonstrated better tolerance among patients with knee OA. Curcumin can be an alternative treatment option in the patients with knee OA who are intolerant to the side effects of non-steroidal anti-inflammatory drugs.