Month: March 2020
Mytonomy Launches Custom Patient Messaging Platform to Triage Urgent Visits
Aren’t ready to telehealth, but need a way to triage inbound calls and potential urgent visits through phone and video? Then Mytonomy, the SaaS company and film studio specializing in virtual care, might be a good choice. The company announced they have a custom messaging platform for its Patient ExperienceRead
MDTech Lanches Video-Enabled Telemedicine
As part of MDTech’s enterprise secure text messaging application, MDMessage, MDTech recently developed video-enabled telemedicine functionality. This capability is available as part of the MDMessage standalone solution or as can be combined with MDTech’s mobile charge capture solution, MDCoder. MDMessage simplifies and secures enterprise communications while MDCoder provides an automatedRead
Lumina Health Launches Telehealth No-Cost Webinar
Lumina Health Partners is launching a telehealth no-cost webinar for healthcare providers to guide them in launching telehealth services. This webinar, taking place on March 26 at 10 a.m. CT will offer practical solutions on establishing telehealth services as well as information on how to properly document, code and billRead
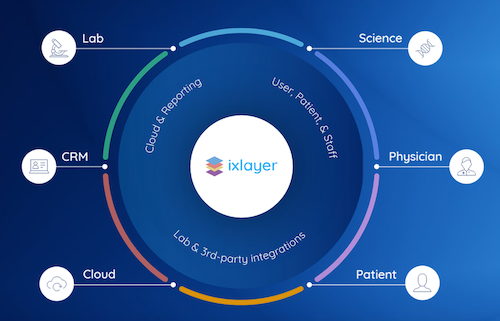
Ixlayer Launches COVID-19 Clinical Testing Platform for in Clinic and Remote Testing
Ixlayer Launches COVID-19 Clinical Testing Platform to Power Labs, and Health Systems for in Clinic and Remote Testing The ixlayer COVID-19 Clinical Test can plug into any health system and lab within 48 hours – enabling labs to digitize ordering and produce real time results delivery while also streamlining healthcareRead
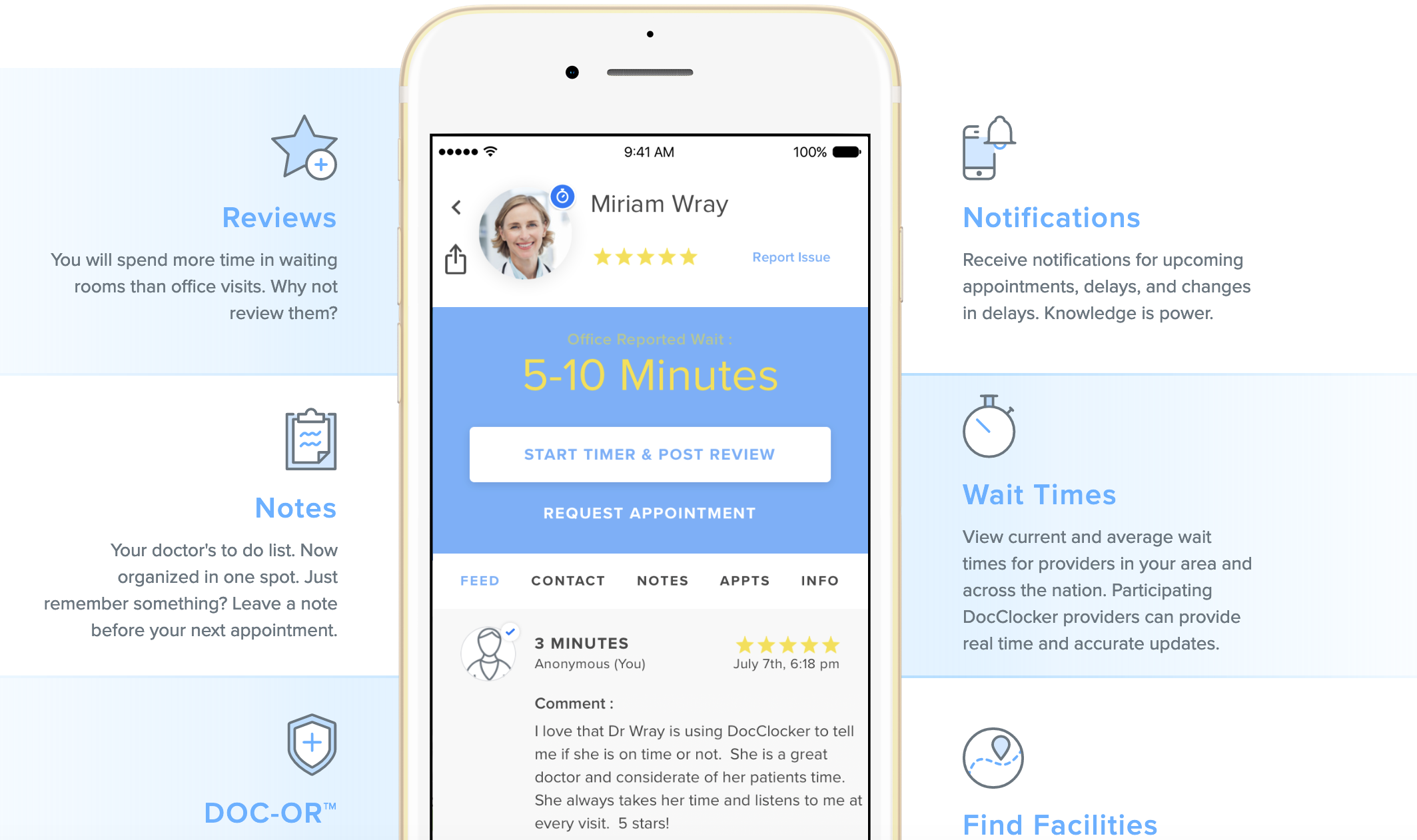
Reducing Waiting Room Exposure to COVID-19
Do you need to reduce waiting time exposure to COVID-19 (coronavirus) and manage your patient wait times? DocClocker®, developed by Fast Pathway, enables medical facilities to provide patients with real-time wait time reporting, which is preventing the spread of COVID-19 nationwide by enabling patients to avoid long waits in medicalRead
Novacyt Achieves US FDA Emergency Use Authorization for COVID-19 Diagnostic Test
Novacyt, an international specialist in clinical diagnostics, is pleased to announce that the US Food and Drug Administration (FDA) has issued an Emergency Use Authorization (EUA) for its COVID-19 diagnostic test. As a result, hospitals and laboratories in the US will be able to use the test for clinical diagnosisRead
CTK Biotech: Coronavirus Test Kits
With cases of coronavirus COVID-19 disease rising at an unprecedented rate, CTK Biotech, in collaboration with its partners in China, has launched two diagnostic coronavirus tests kits, both a real time PCR based nucleic acid detection kit and a serological rapid screening test. Click here to contact CTK about internationalRead
Yes, Friends, the Medical Industry Can Be Transformed: The VA’s Whole Health System of Care at 2 Years
That there should be a huge transformational drama underway in the US medical industry is a desire and dream that powers the integrative health movement and the work of many others. Those who might enjoy a sort of reality TV examination of not the fantasy but the multi-dimensional actuality ofRead
Avoid Junk Food to see Benefits of Plant-based Diets
Plant-based diets are becoming more popular in many areas of the world, but the health benefits of this dietary pattern may depend largely on the specific foods consumed. A new study being presented at the American College of Cardiology’s Annual Scientific Session Together with World Congress of Cardiology (March 28-30,Read
Editorial: Public Health Leadership Paramount to Emerging Coronavirus Pandemic
In this editorial, academic health leaders in Florida and Wisconsin are calling for leadership from Anthony S. Fauci, M.D., director of the United States National Institute of Allergy and Infectious Diseases, to take charge of the coronavirus pandemic. For decades, public health officials have directed the containment of emerging pandemicsRead